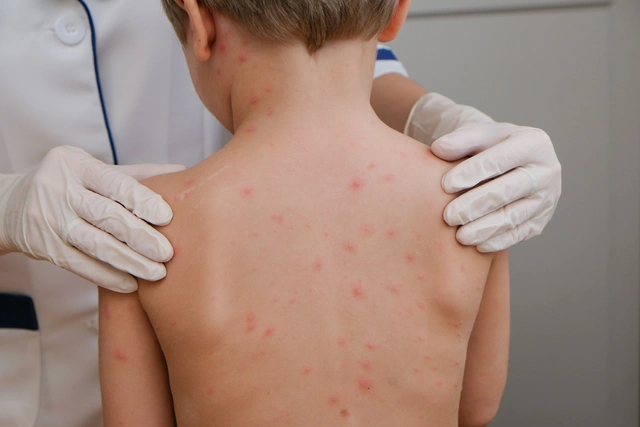
Varicella is an infectious disease caused by the Varicella Zoster Virus (VZV), which belongs to the Herpesviridae family — a group of viruses capable of causing disease in humans.
Each year, varicella cases typically begin to appear from late December and increase significantly in the early months of the year, peaking between February and May. This period coincides with increased group activities and crowded environments for children.
According to statistics from the Pasteur Institute in Ho Chi Minh City, approximately 90% of varicella cases occur in children aged 2 to 7 years.
Due to its high transmissibility, children who attend school and are frequently exposed to crowded settings face a higher risk of infection.
Potential Complications of Varicella
Varicella can lead to complications, particularly in children with weakened immunity or when care is inadequate, including:
– Skin infections, impetigo, and secondary bacterial infections
– Viral pneumonia
– Encephalitis and cerebellitis
– Dehydration resulting from high fever, poor appetite, and reduced fluid intake
In the long term, children may also be at increased risk of developing herpes zoster (shingles)—a late reactivation of the varicella virus that can occur many years after the initial infection. Shingles may lead to complications such as neuropathic pain, corneal ulceration, and vision loss.
Health experts recommend vaccination as the most proactive, safe, and effective method for preventing varicella. Vaccination reduces the risk of infection, limits complications, and protects children throughout the outbreak season. Children aged 12 months and older are advised to receive early vaccination to establish active immunity, thereby reducing the risk of disease and associated complications.
Currently, varicella vaccines have undergone significant improvements, including the second-generation MAV/06 strain varicella vaccine manufactured by GC Biopharma (South Korea), which offers several notable advantages:
– High and durable immunogenicity, with a minimum potency of ≥ 3,800 PFU
– Antibiotic-free formulation, reducing the risk of hypersensitivity reactions in children with allergic predispositions
– WHO Prequalification (WHO-PQ) certification for quality and safety, with experts recommending early vaccination to support long-term health protection
– Cost-effective pricing, supporting expanded immunization coverage
BARYCELA is competitively priced, enabling vaccination centers to more easily provide services to diverse population groups. Affordable pricing encourages parents to vaccinate children earlier, thereby increasing vaccine coverage, reducing outbreak risk, and lowering the overall treatment burden within the community.
Recommended Vaccination Schedule
Children aged 12 months to 12 years:
– Dose 1: From 12 months of age
– Dose 2: At least 3 months after the first dose, or as a booster between 4–6 years of age
Early varicella vaccination serves as a vital immune shield, helping children stay healthy, enjoy a carefree childhood, and remain protected against long-term disease risks.
NAVIVA GROUP is the authorized distributor of the second-generation MAV/06 strain varicella vaccine, BARYCELA Inj. (GC Biopharma – South Korea), in Vietnam. With more than 21 years of experience in vaccines and biological products, NAVIVA GROUP collaborates with healthcare facilities nationwide to enhance access to safe, effective, and high-quality vaccines for the community.
Source: Lao Dong Newspaper