I. VARICELLA
1. Causative virus
Causative agent: Varicella-Zoster virus (VZV) – MAV/06 strain, belonging to the Herpesviridae family, Alphaherpesvirinae subfamily.
Characteristics: An enveloped virus with a double-stranded DNA (dsDNA) genome.
Mode of transmission: Spread via respiratory droplets or direct contact with vesicular fluid. Notably, the virus can be transmitted even before the onset of rash
2. Symptoms and complications
Incubation period: Lasts from 10 to 21 days.
Symptoms: Fever, fatigue, and loss of appetite, followed by a rash with vesicular lesions appearing in successive crops. The rash typically begins on the face, chest, and back before spreading across the body. At any given time, multiple lesion stages may be present simultaneously, including macules, papules, vesicles, and crusts.
Serious complications: Without appropriate care, varicella may lead to complications such as pneumonia, encephalitis, or skin infections. In the long term, the virus can remain latent in the sensory ganglia and later reactivate as herpes zoster (shingles), particularly in older age or immunocompromised individuals.
3. Diagnosis based on clinical and paraclinical findings
Clinical diagnosis: Erythematous rash with vesicular lesions appearing in multiple crops and at different stages simultaneously (macules, papules, vesicles, crusts). The rash spreads from the face, chest, and back to the entire body, often accompanied by intense pruritus.
Paraclinical diagnosis: Serology: anti-VZV IgM (marker of acute infection); PCR: detection of VZV DNA in vesicular fluid or blood; Direct fluorescent antibody (DFA) testing from skin lesion samples.
4. Treatment
Symptomatic treatment, anti-pruritic management, and proper skin hygiene; severe cases may be treated with acyclovir.
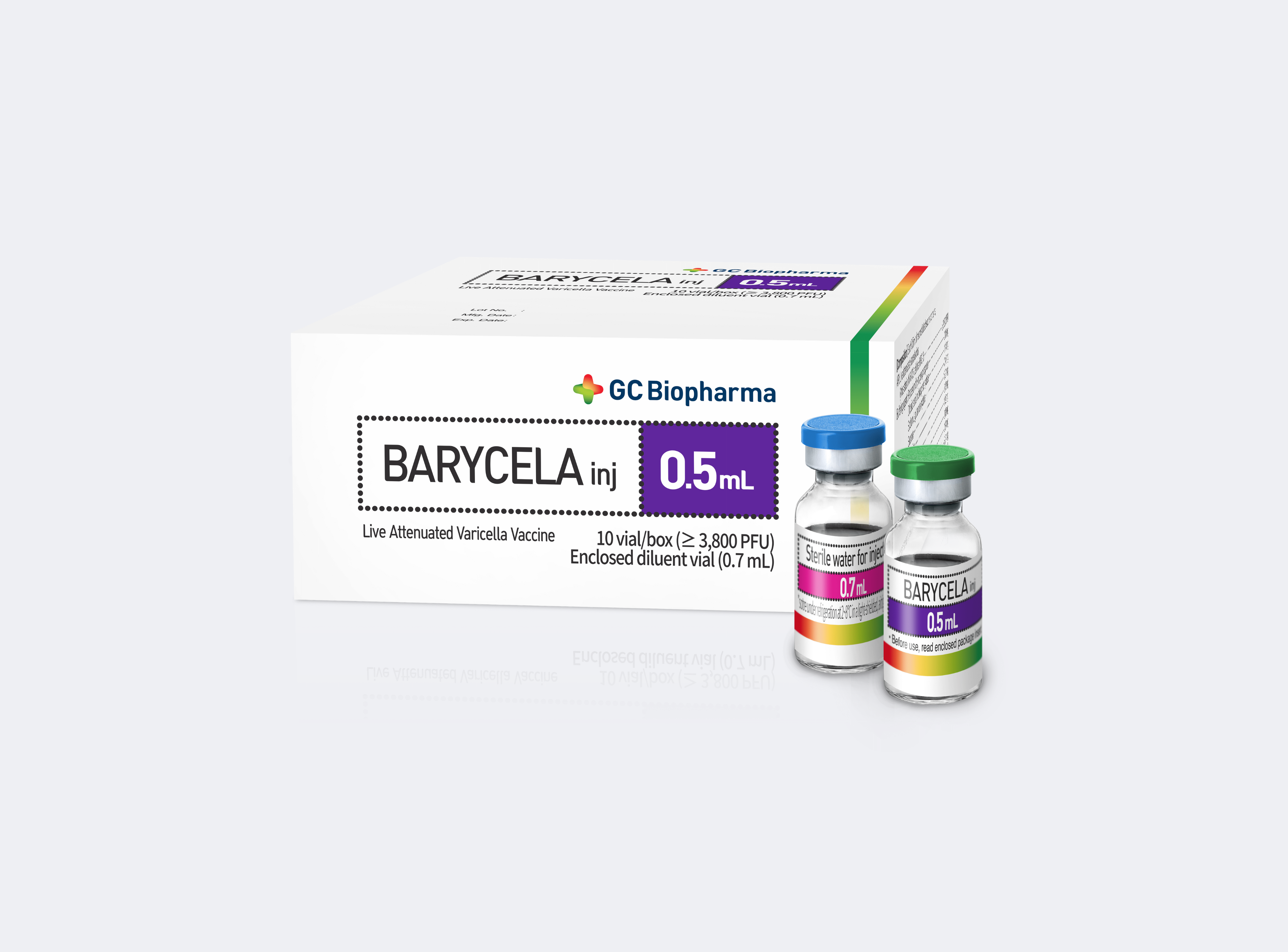
5. BARYCELA VACCINE
BARYCELA inj. is a live attenuated varicella vaccine derived from the Varicella-Zoster virus (attenuated MAV/06 strain), designed to induce an immune response against varicella. The BARYCELA inj. varicella vaccine is manufactured in compliance with Good Manufacturing Practice (GMP), ensuring international quality standards.
Detailed vaccine information:
| Vaccine | BARYCELA
|
| Manufacturer | GC Biopharma Corp., Republic of Korea |
| Composition | Lyophilised vaccine: ✔ Live attenuated varicella virus (Strain: MAV/06, cell line: MRC-5) ≥ 3,800 PFU ✔ Potassium dihydrogen phosphate 0.06 mg ✔ Dibasic sodium phosphate hydrate 1.14 mg ✔ Sodium L-glutamate hydrate 0.40 mg ✔ Sucrose 18.21 mg ✔ L-cysteine 0.18 mg ✔ Glycine 1.82 mg ✔ Disodium edetate hydrate 0.18 mg ✔ Urea 0.87 mg ✔ Gelatin 8.74 mg Diluent: Water for injection: 0.7 mL |
| Vaccine type | Live attenuated |
| Storage | 2–8°C |
| Shelf life | 24 months |
| Vaccination schedule | Dose 1: typically administered from 12 months of age. Dose 2: administered approximately 3 months after Dose 1 to enhance immunogenicity. |
| Route of administration | Subcutaneous |
| Injection site | Young children: anterolateral thigh
Older children: deltoid region (upper arm) |
| Dose | 0.5 mL/dose |
| Indication | Children aged 12 months to 12 years |
| Contraindications | ✔ Severe allergy (anaphylaxis) to any component of the vaccine (e.g. gelatin). ✔ Immunocompromised individuals (congenital disorders, advanced HIV infection, undergoing chemotherapy/radiotherapy, or receiving high-dose immunosuppressive therapy). ✔ Pregnant women or those planning pregnancy within 3 months after vaccination. ✔ Individuals with acute high fever or severe systemic illness (vaccination should be deferred, not an absolute contraindication). ✔ History of severe reaction to a previous dose. ✔ Untreated active tuberculosis. |
Quality control:
| ✔ Seed lot | ✔ Bulk intermediate stage |
| ✔ Cell substrate | ✔ Final bulk |
| ✔ Virus harvest stage | ✔ Final product |
| ✔ Purification |
Final product testing:
| ✔ Appearance | ✔ Residual moisture |
| ✔ Reconstitution time | ✔ Residual BSA content |
| ✔ pH | ✔ Urea content |
| ✔ Potency: Varicella | ✔ Sterility |
| ✔ Identification: Varicella | ✔ Mycoplasma |
BARYCELA vaccine
Source: WHO, CDC, KFDA (Republic of Korea), MSD, Infectious Diseases Textbook – Medical Publishing House.
Dr. Phan Thi Kim Linh, Quality System Management Department
According to: NATIONAL INSTITUTE FOR CONTROL OF VACCINES AND BIOLOGICALS